Probiotics and Gout: New Research
Defining a Healthy Diet: Evidence for the Role of Contemporary Dietary Patterns in Health and Disease
Hellas Cena
1Laboratory of Dietetics and Clinical Nutrition, Department of Public Health, Experimental and Forensic Medicine, University of Pavia, 27100 Pavia, Italy
2Clinical Nutrition and Dietetics Service, Unit of Internal Medicine and Endocrinology, ICS Maugeri IRCCS, 27100 Pavia, Italy
1,2,*, Philip C Calder
3Human Development and Health, Faculty of Medicine, University of Southampton, Southampton SO16 6YD, UK
4NIHR Southampton Biomedical Research Centre, University Hospital Southampton NHS Foundation Trust and University of Southampton, Southampton SO16 6YD, UK
3,4
Abstract
The concept of a healthy diet is continuously evolving, reflecting our expanding understanding of how various foods, essential nutrients, and other dietary components impact health and disease. A substantial and growing body of evidence confirms that consuming specific nutrients, particular food groups, or adopting comprehensive dietary patterns can profoundly influence health and help prevent common non-communicable diseases (NCDs). Eating habits that prioritize health-promoting foods while limiting less healthy options are fundamental to traditional regional diets, such as the Mediterranean diet, and form the basis of structured dietary patterns designed to mitigate disease risk, including the Dietary Approaches to Stop Hypertension (DASH) and Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) diets.
Compared to a typical Western diet, these healthier alternatives consistently feature a higher intake of plant-based foods, including fresh fruits, vegetables, whole grains, legumes, seeds, and nuts. Conversely, they significantly reduce consumption of animal-based foods, particularly fatty and processed meats. To clarify the contemporary understanding of a "healthy diet," this review examines the characteristics and supporting clinical and epidemiological data for various diets recognized for their disease prevention and health-promoting benefits. Collectively, evidence from extensive epidemiological studies and rigorous clinical trials demonstrates that these dietary patterns effectively lower the risks of prevalent NCDs, such as cardiovascular disease and cancer.
1. Introduction
Non-communicable diseases (NCDs), encompassing conditions like cardiovascular disease, cancer, chronic respiratory diseases, diabetes, obesity, and cognitive impairment, represent a leading cause of global mortality and disability. These conditions affect populations across both developed and developing nations [1]. While genetic predispositions and environmental factors contribute to NCD risk, modifiable lifestyle choices, especially at the individual level, play a significant role [2,3,4]. Dietary habits, for instance, are known to influence the risk of developing hypertension, hypercholesterolemia, overweight/obesity, and chronic inflammation. These factors, in turn, escalate the risk for severe health issues such as cardiovascular disease, diabetes, and various cancers [5]. Indeed, the notable increase in chronic NCDs is causally linked to global dietary patterns that are increasingly resembling the Western diet, characterized by high levels of fatty and processed meats, saturated fats, refined grains, excessive salt, and sugars, alongside a notable deficiency in fresh fruits and vegetables [6].
Recognizing the pivotal role of diet in disease risk, the World Health Organization (WHO) Global Action Plan for the Prevention and Control of Noncommunicable Diseases includes strategies to address unhealthy dietary patterns. This initiative is part of broader efforts to mitigate behavioral risk factors, which also include physical inactivity, tobacco use, and harmful alcohol consumption [1]. The WHO's dietary recommendations advise balancing energy intake, limiting saturated and trans fats in favor of unsaturated fats, increasing fruit and vegetable consumption, and restricting sugar and salt intake. Many of these dietary objectives are inherent in regional diets like the Mediterranean diet [7] or are integral components of evidence-based dietary plans specifically designed to reduce disease risk, such as the Dietary Approaches to Stop Hypertension (DASH) [8] and the Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) [9] diets. This narrative review aims to enhance understanding of what constitutes a "healthy diet" by detailing the characteristics and supporting clinical and epidemiological data for diets that align with WHO guidance and have demonstrated positive impacts on health and disease prevention.
2. Components of a Healthy Diet and Their Benefits
A truly healthy diet ensures that macronutrients are consumed in appropriate proportions to meet energetic and physiological demands without excess, while also supplying sufficient micronutrients and hydration for optimal bodily function [10]. Macronutrients—carbohydrates, proteins, and fats—are vital for providing the energy required for all cellular processes and daily activities [11]. Micronutrients, comprising vitamins and minerals, are needed in comparatively small quantities but are indispensable for normal growth, proper development, efficient metabolism, and overall physiological health [12,13]. Balancing these elements is crucial for long-term well-being.
Carbohydrates serve as the body's primary energy source and are abundantly found in grains, fruits, legumes, and vegetables [14]. For maximal health benefits, whole grains are preferable to processed grains. The latter undergo milling, a process that removes the germ and bran, significantly reducing their fiber and micronutrient content [15]. Extensive meta-analyses of prospective cohort studies have consistently linked increased whole-grain intake to a reduced risk of coronary heart disease, stroke, cardiovascular disease, and cancer. Furthermore, higher whole-grain consumption is associated with decreased mortality from all causes, including cardiovascular disease, cancer, respiratory disease, diabetes, and infectious disease [15,16,17]. Fresh fruits and vegetables, beyond providing energy, supply dietary fiber, which promotes satiety and positively influences gastrointestinal function, cholesterol levels, and glycemic control [18]. These foods are also rich sources of phytochemicals, such as polyphenols, phytosterols, and carotenoids. These bioactive compounds are believed to confer many of the health benefits associated with fruit and vegetable consumption [19]. While the precise mechanisms of these phytochemicals are still being investigated, they are known to exhibit antioxidative properties and play roles in regulating nuclear transcription factors, fat metabolism, and inflammatory mediators. For instance, flavonoids have been shown to enhance insulin secretion and reduce insulin resistance, suggesting benefits in managing obesity and diabetes [20]. Additionally, polyphenols interact bidirectionally with gastrointestinal microbiota, fostering beneficial gut bacteria and being metabolized by these bacteria into more active compounds [20]. Studies consistently show an inverse correlation between fruit and vegetable intake and the risk of NCDs, including hypertension [21], cardiovascular disease [22,23], chronic obstructive pulmonary disease [24], lung cancer [25], and metabolic syndrome [26].
Dietary proteins offer both energy and essential amino acids, which the human body cannot produce on its own. Proteins are sourced from both animals (meat, dairy, fish, eggs) and plants (legumes, soy products, grains, nuts, seeds) [27]. Animal-based proteins are often considered richer due to their comprehensive amino acid profiles, high digestibility, and greater bioavailability. However, these sources also contain saturated fatty acids, which are linked to cardiovascular disease, dyslipidemia, and certain cancers [28]. Notably, red and processed meats have been associated with an increased risk of colorectal cancer [28,29]. Furthermore, animal-derived proteins can increase the body's dietary acid load, potentially shifting its acid-base balance towards acidosis [30,31]. This elevated metabolic acid load has been implicated in insulin resistance, impaired glucose homeostasis, and the formation of urinary calcium stones [30,31]. Adequate dietary protein intake is essential throughout life for maintaining lean body mass. In older adults, protein plays a critical role in preventing age-related muscle loss [32], preserving bone density, and reducing fracture risk [33]. For elderly individuals with insufficient dietary protein, amino acid supplementation can enhance strength and functional status [34].
Fats, or lipids, are crucial as primary structural components of cellular membranes and as concentrated sources of cellular energy [35]. Dietary fats are categorized into four main types: monounsaturated, polyunsaturated, saturated, and trans fats, with most foods containing a mixture of these [35]. Unsaturated fats are commonly found in fish, various plant-derived oils, nuts, and seeds, while animal products and some plant oils contribute a higher proportion of saturated fats [35,36]. Trans fats in foods primarily result from the processing of vegetable oils, though small amounts naturally occur in animal products from ruminants like cows, sheep, and goats [35,36]. Among these, unsaturated fats are consistently linked to reduced cardiovascular and mortality risks. Conversely, trans fats and, to a lesser extent, saturated fats, are associated with negative health impacts, including increased mortality [36,37]. Two families of polyunsaturated fatty acids, omega-3 and omega-6, are classified as essential fatty acids because the body cannot produce them and must obtain them from the diet [10]. Omega-3 fatty acids, particularly eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), have been extensively studied for their wide-ranging health benefits, including cardioprotection, cognitive decline prevention, inflammation reduction, muscle mass maintenance, and improved systemic insulin resistance [38,39,40]. Seafood, especially oily fish, is a primary source of EPA and DHA, and supplements are widely available for those who do not meet recommended intakes through diet alone [41,42]. Alpha-linolenic acid, the main plant-based omega-3 fatty acid, can be found in nuts, some seeds, and certain plant oils [43].
While required in much smaller quantities compared to macronutrients, micronutrients are fundamental for normal growth, proper metabolism, optimal physiological functioning, and maintaining cellular integrity [12,13]. The modern Western diet, with its shift from whole foods to processed and refined options, has unfortunately diminished the micronutrient quality of typical food intake [44]. Deficiencies in vitamins and minerals have been implicated in accelerated cellular aging and the onset of late-life diseases, as insufficient supply can lead to chronic metabolic disruption. Consistent with these observations, adequate dietary intake or supplementation with micronutrients possessing antioxidant properties (e.g., vitamins A, C, and E, copper, zinc, and selenium) has been proposed as a strategy to reduce the risk and progression of age-related diseases [45].
Water is the body's most abundant component, forming the majority of lean body mass and total body weight [13]. Beyond providing essential hydration, water also acts as a crucial carrier for micronutrients, including trace elements and electrolytes [46,47]. Drinking water can supply a significant portion, up to 20%, of the daily recommended intake of vital minerals like calcium and magnesium [47]. Despite its fundamental importance, our understanding of specific water requirements and its comprehensive effects on health and disease remains somewhat limited. However, the global increase in the consumption of high-calorie beverages has renewed focus on the critical role of pure water in maintaining overall health and preventing illness [46].
3. Common Health-Promoting Dietary Patterns
Based on our current understanding of nutritional requirements and their profound health impacts, healthy dietary patterns generally share common characteristics. They are typically rich in health-promoting foods such as plant-based options, fresh fruits and vegetables, antioxidants, soy products, nuts, and sources of omega-3 fatty acids. Concurrently, these diets are low in saturated and trans fats, animal-derived proteins, and added or refined sugars [48]. Such patterns may arise organically from regional traditions and local food availability, as seen with the traditional Mediterranean and Asian diets. Alternatively, they can be meticulously developed through research focusing on nutrient intake and subsequent health outcomes, exemplified by the DASH [8] and MIND [9] diets. Despite their diverse origins, these dietary patterns exhibit several common traits, as illustrated below.
Figure 1.
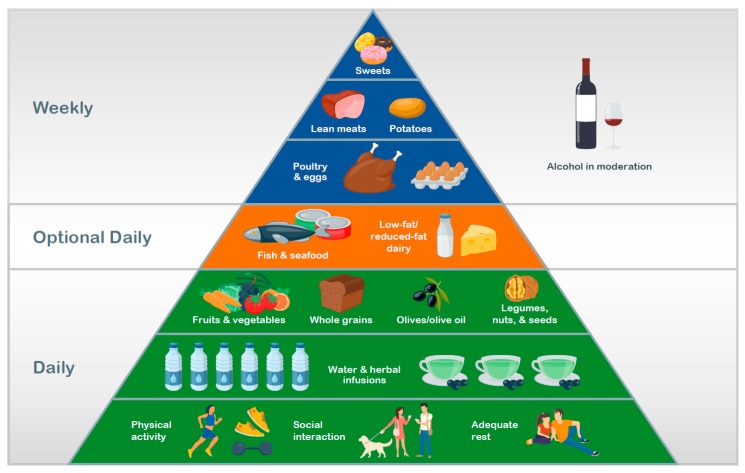
A generalized healthy diet and lifestyle pyramid.
Table 1.
Comparison of nutritional/lifestyle components among different healthy diet options.
| Dietary Component | Recommended Servings |
|---|---|
| Mediterranean [7] | DASH [49] a |
| Fruits | 1–2/meal |
| Vegetables | ≥ 2 servings/meal |
| Whole grains | 1–2 servings/meal |
| Dairy | Low-fat: 2 servings/day |
| Nuts, seeds, and legumes | Olives/nuts/seeds: 1–2 servings/dayLegumes: ≥ 2 servings/week |
| Beef, pork, ham, lamb, veal, poultry | Red meat: < 2 servings/weekProcessed meat: ≤ 1 servings/weekWhite meat: 2 servings/week |
| Poultry: ≥ 2 servings/week | Poultry: ≤ 300 g/week |
| Fish/seafood | ≥ 2 servings/week |
| Fats, oils, and salad dressing | Olive oil: 1–2 servings/meal |
| Sweets | ≤ 2 servings/week |
| Other | Eggs: 2–4 servings/weekPotatoes: ≤ 3 servings//week |
| Alcohol | Wine: in moderation |
| a Recommendations shown here are based on a 2000 calorie per day eating plan. b Contribution of total fat and quality of fat from cheese to stay within the recommended daily intake. |
3.1. Mediterranean Diet
The Mediterranean diet, a renowned dietary pattern, draws its principles from the traditional eating habits of countries bordering the Euro-Mediterranean Sea. Beyond merely defining food types and their proportional intake, it emphasizes a holistic approach to eating. This includes mindfulness about food sourcing (prioritizing sustainability and eco-friendliness), preparation methods, and the social ritual of sharing meals. Crucially, it also integrates broader lifestyle considerations such as regular physical activity and adequate rest [7]. While a core framework exists, regional variations are common, reflecting the emphasis on incorporating traditional and local food products. The daily foundation of the Mediterranean diet comprises whole-grain cereals like bread, pasta, and couscous, rich in fiber, alongside an abundance of diverse fruits and vegetables, offering a spectrum of micronutrients, fiber, and phytochemicals (Table 1) [7,9,49,50,51,52].
Olive oil is the primary source of dietary fats, complemented by olives, nuts, and seeds. Dairy products, preferably low-fat options like yogurt or fermented cheese, are recommended daily in moderation to supply calcium, essential for bone and heart health. Water (1.5–2.0 L/day, or approximately 8 glasses) is advocated as the main source of hydration, with wine and other fermented alcoholic beverages generally permitted in moderation, ideally consumed with meals. Protein sources predominantly include fish, white meat, and eggs, with red and processed meats consumed less frequently and in smaller portions. Legumes are also a favored plant-based protein source within this diet [7]. The health benefits of the Mediterranean diet were first noted in 1975 by Ancel Keys, who observed reduced cardiovascular disease risk among populations adhering to this nutritional model [53]. Since then, research has continually illuminated its beneficial effects on numerous NCDs and related health metrics, including cardiovascular and cerebrovascular disease [54], cancer [55], glycemic control [56], and cognitive function [57,58].
Despite the retraction of a pivotal intervention study (PREDIMED) that evaluated the Mediterranean diet for primary prevention of cardiovascular disease, due to randomization irregularities [59], a subsequent re-analysis, meticulously adjusting for these issues, affirmed the consistent positive impact of adhering to a Mediterranean diet supplemented with olive oil or nuts, compared to a reduced-fat control diet [59]. Sub-studies of PREDIMED have further demonstrated that, relative to a low-fat control diet, the Mediterranean diet, when supplemented with olive oil or nuts, is associated with a 30% reduced risk of major cardiovascular events [59]. It also led to significant reductions in systolic and diastolic blood pressure (5.8–7.3 mmHg and 3.3–3.4 mmHg, respectively) [60]. Additionally, cardiovascular factors such as mean internal carotid artery intima-media thickness (−0.084 mm; p < 0.05) and maximum plaque height (−0.091 mm; p < 0.05) showed improvement with the Mediterranean diet supplemented with nuts [61]. Greater intake of polyphenols, beneficial phytochemicals abundant in fruits, vegetables, tea, olive oil, and wine, correlated with a 36% reduced risk of hypertension (p = 0.015) [62] and improvements in inflammatory biomarkers linked to atherosclerosis (e.g., interleukin [IL]-6, tumor necrosis factor-alpha, soluble intercellular adhesion molecule-1, vascular cell adhesion molecule-1, and monocyte chemotactic protein-1; p < 0.05 for each), as well as increased high-density lipoprotein cholesterol (HDL-C; p = 0.004) [62,63].
3.2. Dietary Approaches to Stop Hypertension (DASH)
The DASH diet, named after the Dietary Approaches to Stop Hypertension study, originated from research specifically investigating the impact of dietary patterns on blood pressure [8]. Participants consuming a diet rich in fruits, vegetables, and low-fat dairy, while reducing saturated and total fat and cholesterol, experienced significantly greater blood pressure reductions compared to those on a typical American control diet (difference in systolic/diastolic blood pressure: −5.5/−3.0 mmHg; p < 0.001) or a diet high in fruits and vegetables but with fewer snacks and sweets (−2.7/−1.9 mmHg; p ≤ 0.002). All three diets maintained a sodium content of 3 grams per day. A subsequent study, DASH-Sodium, explored the DASH diet or a control diet in conjunction with varying sodium levels (high, intermediate, and low), finding that the DASH diet consistently reduced systolic blood pressure across all sodium phases (high: −5.9 mmHg; p < 0.001; intermediate: −5.0 mmHg; p < 0.001; low: −2.2 mmHg; p < 0.05) [64].
The DASH diet also significantly lowered diastolic blood pressure compared to the control diet during high (−2.9 mmHg; p < 0.001) and intermediate (−2.5 mmHg; p < 0.01) sodium intake phases, though not during the low intake phase (−1.0 mmHg). While reducing sodium intake alone also significantly lowered blood pressure in the control group (p < 0.05), the low sodium phase of the DASH diet achieved even more significant decreases in systolic/diastolic blood pressure of −8.9/−4.5 mmHg (p < 0.001 for both) when compared to the high sodium intake phase of the control diet. Subsequent controlled trials have generally affirmed these findings, extending the known benefits of the DASH diet to include improvements in other cardiovascular risk factors and comorbidities, such as reduced low-density lipoprotein cholesterol (LDL-C), total cholesterol, mitigated overweight/obesity, and enhanced insulin sensitivity [65,66,67,68]. The DASH diet has also been linked to reductions in adverse outcomes including cardiovascular disease, coronary heart disease, stroke, heart failure, metabolic syndrome, and diabetes, with improved pregnancy outcomes in women with gestational diabetes [68,69,70,71,72].
Meta-analyses of studies employing the DASH diet reveal significant reductions in LDL-C by −0.1 mmol/L (p = 0.03) [65,68], total cholesterol by −0.2 mmol/L (p < 0.001) [65,68], body weight by −1.42 kg (p < 0.001) [66,68], and fasting insulin by −0.15 μU/mL (p < 0.001) [65,66,67,68]. Adherence to the DASH diet is associated with a 20% reduction in cardiovascular disease risk, a 19% reduction in stroke risk, and a 29% reduction in heart failure risk (p < 0.001 for each) [69,71]. The overall risk of diabetes is reduced by 18% [68], and children and adolescents with higher DASH scores—indicating greater consumption of fruits, vegetables, nuts, legumes, low-fat dairy, and whole grains—demonstrated a 64% lower risk of developing metabolic syndrome compared to those with the lowest scores (p = 0.023) [71]. Furthermore, rates of cesarean section decreased by 47% [72], the incidence of macrosomia (birth weight > 4000 g) fell from 39% to 4% (p = 0.002) [70], and significantly fewer women with gestational diabetes on the DASH diet required insulin therapy (23%) compared to the control diet (73%; p < 0.0001) [70]. The dietary pattern derived from the DASH study emphasizes a wide variety of vegetables (including colorful, starchy, and legume varieties), fruits, fat-free or low-fat dairy products, whole grains, and diverse protein sources such as seafood, lean meats, eggs, legumes, nuts, seeds, and soy (Table 1) [49]. Limited consumption of added sugars (<10% of daily calories), saturated fats (<10% of daily calories), sodium (<2300 mg/day), and alcohol (≤1 drink per day for women, ≤2 for men) is strongly advised. Further reductions in blood pressure may be achieved by even lower sodium intake, although practical challenges can limit the feasibility of reaching 1200 mg or less per day [49].
3.3. Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND)
The MIND diet represents a strategic fusion of key elements from both the Mediterranean and DASH diets, specifically formulated to promote cognitive health as individuals age [9]. Individually, both the Mediterranean and DASH diets have been linked to positive cognitive outcomes, including the prevention of cognitive decline or impairment and improved cognitive performance [73,74,75]. Two high-quality cohort studies have demonstrated significant associations between adherence to the MIND diet and a 53% lower risk for developing Alzheimer’s disease (p = 0.002 for linear trend) [50]. Furthermore, higher adherence to the MIND diet was associated with slower declines in overall cognitive functioning and within specific cognitive domains (e.g., episodic, semantic, and working memory, as well as perceptual speed and organization), translating to cognitive function equivalent to being 7.5 years younger [50,76].
Notably, even moderate adherence to the MIND diet was associated with a 35% reduction in Alzheimer’s disease risk compared to the lowest adherence group (p = 0.002 for linear trend). In contrast, the Mediterranean and DASH diets required high adherence to demonstrate statistically significant risk reductions of 54% and 39% respectively, highlighting the unique effectiveness of the MIND diet at lower adherence levels [50]. The MIND diet specifically focuses on increasing the intake of fresh fruits and vegetables, emphasizing brain-healthy foods such as green leafy vegetables, nuts, berries, beans, whole grains, fish, poultry, olive oil, and moderate wine consumption (Table 1) [9,50]. It also distinctively limits foods considered detrimental to brain health, including red meats, butter/margarine, cheese, pastries, sweets, and fried or fast food [9]. This specificity in recommending and limiting certain food types differentiates the MIND diet from its Mediterranean and DASH counterparts [50].
3.4. Nordic Diet
The Nordic diet, in its various forms (e.g., healthy Nordic diet, New Nordic Diet), emerged from a desire to adapt the principles of the Mediterranean, DASH, and other health-promoting diets into a regionally specific dietary pattern. This adaptation focuses on traditional, local Nordic foods, aiming for public appeal, sustainability, and eco-friendliness [77,78]. The overarching tenets of the New Nordic Diet advocate for increased caloric intake from plant sources and reduced intake from animal sources, greater consumption of foods from seas and lakes, and an emphasis on wild countryside foods [78,79]. A generalized Nordic dietary pattern typically includes green leafy and other vegetables, fruits, fish and seafood, potatoes, berries, whole grains (such as wheat, rye, oats, barley), nuts, low-fat dairy products, and oils like rapeseed, sunflower, or soy. It also limits fresh red meat and sugar [78,80]. Specific dietary recommendations, as derived from the NORDIET clinical trial, are detailed in Table 1 [51].
The randomized, controlled NORDIET study compared a healthy Nordic diet with a control diet (participants' usual Western diet) over a 6-week period [77]. The Nordic diet demonstrated significant improvements in participants' lipid profiles, including a 0.98 mmol/L reduction in total cholesterol (p < 0.0001) and a 0.83 mmol/L reduction in LDL-C (p < 0.001). It also lowered systolic blood pressure by 6.6 mmHg (p = 0.008) and enhanced insulin sensitivity, with homeostatic model assessment-insulin resistance decreasing by 0.11 (p = 0.01) when compared to the control diet. Notably, participants on the Nordic diet experienced a 3.0 kg decrease in body weight (p < 0.001) despite being allowed to eat ad libitum.
Subsequent studies using variations of the Nordic diet have consistently reinforced these findings, showing improvements relative to control diets in blood lipid profiles (LDL-C/HDL-C ratio, −0.15; p = 0.046) [81] and inflammation (IL-1 receptor antagonist, −84 ng/L; p < 0.001) [81]. Among patients with metabolic syndrome, there were also improvements in blood pressure (DBP, −4.4 mmHg (p = 0.001), and mean arterial pressure (−4.2 mmHg; p = 0.006) [82], as well as weight loss (−3.22 kg; p < 0.001) [83] and blood pressure reduction (SBP/DBP, −5.13/−3.24 mmHg; p < 0.05) in individuals with obesity [83]. One study even showed baseline blood pressure reductions of −6.9 mmHg (SBP) and −3.2 mmHg (DBP; p < 0.01) [83,84]. Furthermore, a study in children reported that improvements in omega-3 fatty acid status resulting from the Nordic diet were associated with better school performance (p < 0.05) [85]. A systematic review analyzing individual components of the Nordic diet found strong evidence supporting the protective effects of whole grains on type 2 diabetes and cardiovascular disease risk, though evidence for other individual foods within the Nordic diet was less conclusive [86].
3.5. Traditional Asian Diets
While substantial evidence supports the Mediterranean and other European-based diets, traditional regional dietary patterns from other parts of the world, though often adhering to similar healthful principles, have less extensively established links to positive health outcomes. A comprehensive review of the vast array of global regional diets and their associated evidence is beyond the scope of this publication. However, we will briefly consider some Asian-based diets that are particularly relevant to this discussion due to their historical prominence and observed health impacts.
The traditional Korean diet is characterized by its reliance on rice and other whole grains, fermented foods, a wide array of indigenous land and sea vegetables, and proteins derived primarily from legumes and fish rather than red meat. It also incorporates medicinal herbs like garlic, green onions, and ginger, along with sesame and perilla oils [87]. Meals typically consist of multiple small-portion dishes, often prepared with seasonal ingredients and cooked at home, contrasting sharply with the Western diet’s frequent inclusion of fried foods [87]. Epidemiological data indicate a reduced risk of metabolic syndrome (odds ratio [OR]: 0.77; 95% CI: 0.60–0.99), obesity (OR: 0.72; 95% CI: 0.55–0.95), hypertension (OR: 0.74; 95% CI: 0.57–0.98), and hypertriglyceridemia (OR: 0.76; 95% CI: 0.59–0.99) among individuals adhering to traditional Korean dietary patterns [88]. These findings are supported by a controlled clinical trial that explored the impact of a traditional Korean diet versus a control diet ("eat as usual") on cardiovascular risk factors in patients with diabetes and hypertension. In this study, adherence to a traditional Korean diet favorably influenced body composition (body weight, −2.3 kg; body mass index [BMI], −0.83 kg/m²; body fat, −2.2%; p < 0.01), heart rate (−7.1 bpm; p = 0.002), and glycemic control (HbA1c, −0.72%; p = 0.003) [89].
The traditional Chinese diet typically features rice or noodles, soups, various vegetables, steamed breads or dumplings, fruits, soy products, seafood, and moderate amounts of meat [90,91]. Although this diet is generally higher in carbohydrates and lower in fat compared to a Western diet, it does not appear to promote weight gain in healthy, normal-weight Chinese individuals. This observation suggests that carbohydrate restriction may not be a universally applicable intervention for combating obesity and cardiometabolic risk [92]. One 6-week controlled trial demonstrated that 52% of non-Chinese individuals who were overweight or obese and adhered to a traditional Chinese diet experienced a reduction in BMI while preserving lean body mass, compared to 28% of those who followed a Western diet at the 1-year follow-up assessment [93]. In another trial, subjects adhering to a traditional Chinese diet for 6 weeks saw a decrease in BMI of 0.37 kg/m² and a reduction in lean mass of 0.21 kg, whereas those on a Western diet experienced reductions of 0.26 kg/m² in BMI and 0.49 kg in lean body mass, respectively [94]. It is noteworthy that both of these studies imposed a caloric restriction of 1,200 Kcal for both test and control diet groups.
Similar to the Korean diet, the traditional Japanese diet, known as Washoku, emphasizes small portions across multiple components, primarily including rice, fish (often consumed raw), soups, and pickles [95]. Fermented soybean paste (dashi) forms the base of many central soups in the traditional Japanese diet, with other common ingredients including seaweed, various fruits and vegetables, and mushrooms. Cultural eating habits, such as the use of chopsticks, alternating between small dishes throughout a meal, and the umami flavor profile inherent in Japanese food, contribute to enhanced satiety and help prevent overeating. Adherence to a traditional Japanese dietary pattern has been linked to favorable effects on blood pressure among healthy Japanese adults [96]. This aligns with data from the 2012 Japan National Health and Nutrition Survey, which demonstrated that adherence to a traditional Japanese diet was associated with a lower prevalence of hypertension in men, compared to a Western diet or a meat- and fat-based dietary pattern [97]. However, the same study also reported that the traditional Japanese diet was associated with higher diastolic blood pressure in women, and increased waist circumference and BMI in men. Further research is therefore necessary to fully elucidate the comprehensive health impacts of traditional Japanese and other Asian dietary patterns.
4. Additional Factors
While the evidence presented strongly indicates that the described dietary patterns positively influence health metrics and disease outcomes by promoting beneficial food choices and discouraging unhealthy ones, it is important to recognize that a healthy lifestyle is a composite of multiple contributing factors. These dietary patterns, for instance, consistently advocate for adequate hydration, primarily through water or herbal infusions [7,49,51,52]. Beyond just food, a truly healthy lifestyle integrates regular physical activity, fosters social connections, ensures sufficient sleep [7,52], and actively minimizes detrimental elements such as tobacco use, excessive alcohol consumption, prolonged physical inactivity, excessive screen time, and chronic stress.
The crucial role of these non-dietary factors is often visually represented in modern food pyramids. These pyramids typically feature a foundational tier of positive lifestyle factors, upon which subsequent tiers of daily consumption are built. These lower tiers highlight adequate hydration and nutrient-rich, plant-based foods. Animal-derived products (meat, fish, and dairy) and sweets generally occupy the higher tiers of the pyramid, indicating less frequent or infrequent consumption. While the ideal goal is to meet all nutrient requirements through food and water alone, there are circumstances where food-derived nutrient intake might be insufficient. This can occur due to increased physiological needs, highly selective eating habits, or limitations in food security and access to nutritious options [98,99,100]. Consequently, for some individuals, particularly during specific life phases, dietary supplements may become necessary. For example, as people age, the recommended intake of calcium increases to maintain bone mineral density [101], making calcium supplementation potentially crucial for older adults to meet recommended levels. Before initiating any supplementation, a careful assessment of existing dietary intake is vital to prevent exceeding upper tolerability limits and potentially causing adverse effects.
It is important to acknowledge that numerous other traditional regional diets likely offer health benefits similar to those discussed here. However, this review focused specifically on diets supported by evidence from randomized, controlled trials demonstrating their health advantages. For instance, the African Heritage Diet, which emphasizes traditional ingredients, holds significant potential benefits for African American populations, who often face disproportionately higher risks for diet-related chronic diseases [102]. Future research is warranted to thoroughly evaluate the impact of the African Heritage Diet and other diverse regional dietary patterns on overall health and well-being.
5. Conclusions
Healthy diets, whether rooted in long-standing tradition or meticulously designed through scientific inquiry, share a multitude of common features and generally align with the World Health Organization's Global Action Plan for the Prevention and Control of Noncommunicable Diseases. Compared to a typical Western diet, these more healthful alternatives are characterized by a higher abundance of plant-based foods, including fresh fruits, vegetables, whole grains, legumes, seeds, and nuts. Conversely, they entail a reduced intake of animal-based foods, particularly fatty and processed meats. Robust evidence from both epidemiological studies and clinical trials consistently demonstrates that these dietary patterns significantly reduce the risks of a wide array of NCDs, spanning from cardiovascular disease to various forms of cancer. Moving forward, concerted efforts are essential to integrate these healthy dietary and lifestyle choices into daily life within communities worldwide, striving to make healthy eating accessible, achievable, and sustainable for all.
Acknowledgments
Medical writing support was provided by Crystal Murcia, PhD, and Dennis Stancavish, MA, of Peloton Advantage, LLC, an OPEN Health company, and was funded by Pfizer Consumer Healthcare. On 1 August 2019, Pfizer Consumer Healthcare became part of GSK Consumer Healthcare.
Author Contributions
H.C. and P.C.C. contributed to the conception of the work; the acquisition, analysis, and interpretation of data; drafting; and revision of the work. Both have approved the final version for submission and agree to be personally accountable for their contributions and for ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated, resolved, and documented in the literature. All authors have read and agreed to the published version of the manuscript.
Funding
Medical writing support was funded by Pfizer Consumer Healthcare; this research received no other external funding. The APC was funded by Pfizer Consumer Healthcare. On 1 August 2019, Pfizer Consumer Healthcare became part of GSK Consumer Healthcare.
Conflicts of Interest
Hellas Cena received travel reimbursement from Pfizer Consumer Healthcare to attend a discussion meeting prior to drafting the manuscript and acts as a consultant to companies that manufacture or market dietary supplements, including Pfizer Consumer Healthcare. Philip C. Calder received travel reimbursement from Pfizer Consumer Healthcare to attend a discussion meeting prior to drafting the manuscript. Pfizer Consumer Healthcare funded this project, but the company had no role in the design, execution, interpretation, or writing of the paper.
References
- 1.Global Action Plan for the Prevention and Control of Noncommunicable Diseases 2013–2020. World Health Organ; Geneva, Switzerland: 2013.
- 2.Tamimi R.M., Spiegelman D., Smith-Warner S.A., Wang M., Pazaris M., Willett W.C., Eliassen A.H., Hunter D.J. Population attributable risk of modifiable and nonmodifiable breast cancer risk factors in postmenopausal breast cancer. Am. J. Epidemiol. 2016;184:884–893. doi: 10.1093/aje/kww145.
- 3.Yu E., Rimm E., Qi L., Rexrode K., Albert C.M., Sun Q., Willett W.C., Hu F.B., Manson J.E. Diet, lifestyle, biomarkers, genetic factors, and risk of cardiovascular disease in the Nurses’ Health Studies. Am. J. Public Health. 2016;106:1616–1623. doi: 10.2105/AJPH.2016.303316.
- 4.Kolb H., Martin S. Environmental/lifestyle factors in the pathogenesis and prevention of type 2 diabetes. BMC Med. 2017;15:131. doi: 10.1186/s12916-017-0901-x.
- 5.Koene R.J., Prizment A.E., Blaes A., Konety S.H. Shared risk factors in cardiovascular disease and cancer. Circulation. 2016;133:1104–1114. doi: 10.1161/CIRCULATIONAHA.115.020406.
- 6.Cordain L., Eaton S.B., Sebastian A., Mann N., Lindeberg S., Watkins B.A., O’Keefe J.H., Brand-Miller J. Origins and evolution of the Western diet: Health implications for the 21st century. Am. J. Clin. Nutr. 2005;81:341–354. doi: 10.1093/ajcn.81.2.341.
- 7.Bach-Faig A., Berry E.M., Lairon D., Reguant J., Trichopoulou A., Dernini S., Medina F.X., Battino M., Belahsen R., Miranda G., et al. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011;14:2274–2284. doi: 10.1017/S1368980011002515.
- 8.Appel L.J., Moore T.J., Obarzanek E., Vollmer W.M., Svetkey L.P., Sacks F.M., Bray G.A., Vogt T.M., Cutler J.A., Windhauser M.M., et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N. Engl. J. Med. 1997;336:1117–1124. doi: 10.1056/NEJM199704173361601.
- 9.Marcason W. What are the components to the MIND diet? J. Acad. Nutr. Diet. 2015;115:1744. doi: 10.1016/j.jand.2015.08.002.
- 10.Stark C. Guidelines for Food and Nutrient Intake. In: Stipanuk M.H., Caudill M.A., editors. Biochemistry, Physiology and Molecular Aspects of Human Nutrition. 3rd ed. Elsevier Saunders; St. Louis, MO, USA: 2013. pp. 34–47.
- 11.Stipanuk M.H., Caudill M.A. Structure and Properties of the Macronutrients. In: Stipanuk M.H., Caudill M.A., editors. Biochemistry, Physiology and Molecular Aspects of Human Nutrition. 3rd ed. Elsevier Saunders; St. Louis, MO, USA: 2013. p. 49.
- 12.Stipanuk M.H., Caudill M.A. The Vitamins. In: Stipanuk M.H., Caudill M.A., editors. Biochemistry, Physiology and Molecular Aspects of Human Nutrition. 3rd ed. Elsevier Saunders; St. Louis, MO, USA: 2013. pp. 537–539.
- 13.Stipanuk M.H., Caudill M.A. The Minerals and Water. In: Stipanuk M.H., Caudill M.A., editors. Biochemistry, Physiology and Molecular Aspects of Human Nutrition. 3rd ed. Elsevier Saunders; St. Louis, MO, USA: 2013. pp. 719–720.
- 14.Slavin J. Structure, Nomenclature, and Properties of Carbohydrates. In: Stipanuk M.H., Caudill M.A., editors. Biochemistry, Physiology and Molecular Aspects of Human Nutrition. 3rd ed. Elsevier Saunders; St. Louis, MO, USA: 2013. pp. 50–68.
- 15.Benisi-Kohansal S., Saneei P., Salehi-Marzijarani M., Larijani B., Esmaillzadeh A. Whole-grain intake and mortality from all causes, cardiovascular disease, and cancer: A systematic review and dose-response meta-analysis of prospective cohort studies. Adv. Nutr. 2016;7:1052–1065. doi: 10.3945/an.115.011635.
- 16.Aune D., Keum N., Giovannucci E., Fadnes L.T., Boffetta P., Greenwood D.C., Tonstad S., Vatten L.J., Riboli E., Norat T. Whole grain consumption and risk of cardiovascular disease, cancer, and all cause and cause specific mortality: Systematic review and dose-response meta-analysis of prospective studies. BMJ. 2016;353:i2716. doi: 10.1136/bmj.i2716.
- 17.Zong G., Gao A., Hu F.B., Sun Q. Whole grain intake and mortality from all causes, cardiovascular disease, and cancer: A meta-analysis of prospective cohort studies. Circulation. 2016;133:2370–2380. doi: 10.1161/CIRCULATIONAHA.115.021101.
- 18.McRorie J.W., Jr., McKeown N.M. Understanding the physics of functional fibers in the gastrointestinal tract: An evidence-based approach to resolving enduring misconceptions about insoluble and soluble fiber. J. Acad. Nutr. Diet. 2017;117:251–264. doi: 10.1016/j.jand.2016.09.021.
- 19.Probst Y.C., Guan V.X., Kent K. Dietary phytochemical intake from foods and health outcomes: A systematic review protocol and preliminary scoping. BMJ Open. 2017;7:e013337. doi: 10.1136/bmjopen-2016-013337.
- 20.Fraga C.G., Croft K.D., Kennedy D.O., Tomas-Barberan F.A. The effects of polyphenols and other bioactives on human health. Food Funct. 2019;10:514–528. doi: 10.1039/C8FO01997E.
- 21.Li B., Li F., Wang L., Zhang D. Fruit and vegetables consumption and risk of hypertension: A meta-analysis. J. Clin. Hypertens (Greenwich) 2016;18:468–476. doi: 10.1111/jch.12777.
- 22.Gan Y., Tong X., Li L., Cao S., Yin X., Gao C., Herath C., Li W., Jin Z., Chen Y., et al. Consumption of fruit and vegetable and risk of coronary heart disease: A meta-analysis of prospective cohort studies. Int. J. Cardiol. 2015;183:129–137. doi: 10.1016/j.ijcard.2015.01.077.
- 23.Zhan J., Liu Y.J., Cai L.B., Xu F.R., Xie T., He Q.Q. Fruit and vegetable consumption and risk of cardiovascular disease: A meta-analysis of prospective cohort studies. Crit. Rev. Food Sci. Nutr. 2017;57:1650–1663. doi: 10.1080/10408398.2015.1008980.
- 24.Kaluza J., Larsson S.C., Orsini N., Linden A., Wolk A. Fruit and vegetable consumption and risk of COPD: A prospective cohort study of men. Thorax. 2017;72:500–509. doi: 10.1136/thoraxjnl-2015-207851.
- 25.Wang Y., Li F., Wang Z., Qiu T., Shen Y., Wang M. Fruit and vegetable consumption and risk of lung cancer: A dose-response meta-analysis of prospective cohort studies. Lung Cancer. 2015;88:124–130. doi: 10.1016/j.lungcan.2015.02.015.
- 26.Tian Y., Su L., Wang J., Duan X., Jiang X. Fruit and vegetable consumption and risk of the metabolic syndrome: A meta-analysis. Public Health Nutr. 2018;21:756–765. doi: 10.1017/S136898001700310X.
- 27.Lonnie M., Hooker E., Brunstrom J.M., Corfe B.M., Green M.A., Watson A.W., Williams E.A., Stevenson E.J., Penson S., Johnstone A.M. Protein for life: Review of optimal protein intake, sustainable dietary sources and the effect on appetite in ageing adults. Nutrients. 2018;10:360. doi: 10.3390/nu10030360.
- 28.Demeyer D., Mertens B., De Smet S., Ulens M. Mechanisms linking colorectal cancer to the consumption of (processed) red meat: A review. Crit. Rev. Food Sci. Nutr. 2016;56:2747–2766. doi: 10.1080/10408398.2013.873886.
- 29.Meat, Fish and Dairy Products and the Risk of Cancer. [(accessed on 28 May 2019)]; Available online: https://www.wcrf.org/sites/default/files/Meat-Fish-and-Dairy-products.pdf.
- 30.Della Guardia L., Roggi C., Cena H. Diet-induced acidosis and alkali supplementation. Int. J. Food Sci. Nutr. 2016;67:754–761. doi: 10.1080/09637486.2016.1198889.
- 31.Della Guardia L., Thomas M.A., Cena H. Insulin sensitivity and glucose homeostasis can be influenced by metabolic acid load. Nutrients. 2018;10:618. doi: 10.3390/nu10050618.
- 32.Kim J.E., O’Connor L.E., Sands L.P., Slebodnik M.B., Campbell W.W. Effects of dietary protein intake on body composition changes after weight loss in older adults: A systematic review and meta-analysis. Nutr. Rev. 2016;74:210–224. doi: 10.1093/nutrit/nuv065.
- 33.Curneen J.M.G., Casey M., Laird E. The relationship between protein quantity, BMD and fractures in older adults. Ir. J. Med. Sci. 2018;187:111–121. doi: 10.1007/s11845-017-1642-8.
- 34.Cheng H., Kong J., Underwood C., Petocz P., Hirani V., Dawson B., O’Leary F. Systematic review and meta-analysis of the effect of protein and amino acid supplements in older adults with acute or chronic conditions. Br. J. Nutr. 2018;119:527–542. doi: 10.1017/S0007114517003816.
- 35.Brenna J.T., Sacks G.L. Lipid Structure, Nomenclature, and Chemical Properties. In: Stipanuk M.H., Caudill M.A., editors. Biochemistry, Physiology and Molecular Aspects of Human Nutrition. 3rd ed. Elsevier Saunders; St. Louis, MO, USA: 2013. pp. 91–119.
- 36.de Souza R.J., Mente A., Maroleanu A., Cozma A.I., Ha V., Kishibe T., Uleryk E., Budylowski P., Schunemann H., Beyene J., et al. Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: Systematic review and meta-analysis of observational studies. BMJ. 2015;351:h3978. doi: 10.1136/bmj.h3978.
- 37.Ricci C., Baumgartner J., Zec M., Kruger H.S., Smuts C.M. Type of dietary fat intakes in relation to all-cause and cause-specific mortality in US adults: An iso-energetic substitution analysis from the American National Health and Nutrition Examination Survey linked to the US mortality registry. Br. J. Nutr. 2018;119:456–463. doi: 10.1017/S0007114517003889.
- 38.Cederholm T., Salem N., Jr., Palmblad J. Omega-3 fatty acids in the prevention of cognitive decline in humans. Adv. Nutr. 2013;4:672–676. doi: 10.3945/an.113.004556.
- 39.Manuelli M., Della Guardia L., Cena H. Enriching diet with n-3 PUFAs to help prevent cardiovascular diseases in healthy adults: Results from clinical trials. Int. J. Mol. Sci. 2017;18:1552. doi: 10.3390/ijms18071552.
- 40.Buoite Stella A., Gortan Cappellari G., Barazzoni R., Zanetti M. Update on the impact of omega 3 fatty acids on inflammation, insulin resistance and sarcopenia: A review. Int. J. Mol. Sci. 2018;19:218. doi: 10.3390/ijms19010218.
- 41.Calder P.C. Omega-3: The good oil. Nutr. Bull. 2017;42:132–140. doi: 10.1111/nbu.12261.
- 42.Calder P.C. Very long-chain n-3 fatty acids and human health: Fact, fiction and the future. Proc. Nutr. Soc. 2018;77:52–72. doi: 10.1017/S0029665117003950.
- 43.Baker E.J., Miles E.A., Burdge G.C., Yaqoob P., Calder P.C. Metabolism and functional effects of plant-derived omega-3 fatty acids in humans. Prog. Lipid Res. 2016;64:30–56. doi: 10.1016/j.plipres.2016.07.002.
- 44.Ames B.N. Low micronutrient intake may accelerate the degenerative diseases of aging through allocation of scarce micronutrients by triage. Proc. Natl. Acad. Sci. USA. 2006;103:17589–17594. doi: 10.1073/pnas.0608757103.
- 45.Hohn A., Weber D., Jung T., Ott C., Hugo M., Kochlik B., Kehm R., Konig J., Grune T., Castro J.P. Happily (n)ever after: Aging in the context of oxidative stress, proteostasis loss and cellular senescence. Redox Biol. 2017;11:482–501. doi: 10.1016/j.redox.2016.12.001.
- 46.Popkin B.M., D’Anci K.E., Rosenberg I.H. Water, hydration, and health. Nutr. Rev. 2010;68:439–458. doi: 10.1111/j.1753-4887.2010.00304.x.
- 47.World Health Organization . Nutrients in Drinking Water. World Health Organization; Geneva, Switzerland: 2005.
- 48.Pistollato F., Iglesias R.C., Ruiz R., Aparicio S., Crespo J., Lopez L.D., Manna P.P., Giampieri F., Battino M. Nutritional patterns associated with the maintenance of neurocognitive functions and the risk of dementia and Alzheimer’s disease: A focus on human studies. Pharmacol. Res. 2018;131:32–43. doi: 10.1016/j.phrs.2018.03.012.
- 49.Campbell A.P. DASH eating plan: An eating pattern for diabetes management. Diabetes Spectr. 2017;30:76–81. doi: 10.2337/ds16-0084.
- 50.Morris M.C., Tangney C.C., Wang Y., Sacks F.M., Bennett D.A., Aggarwal N.T. MIND diet associated with reduced incidence of Alzheimer’s disease. Alzheimers Dement. 2015;11:1007–1014. doi: 10.1016/j.jalz.2014.11.009.
- 51.Adamsson V., Reumark A., Cederholm T., Vessby B., Riserus U., Johansson G. What is a healthy Nordic diet? Foods and nutrients in the NORDIET study. Food Nutr. Res. 2012;56 doi: 10.3402/fnr.v56i0.18189.
- 52.Asian Heritage Diet. [(accessed on 10 July 2019)]; Available online: https://oldwayspt.org/traditional-diets/asian-heritage-diet.
- 53.Altomare R., Cacciabaudo F., Damiano G., Palumbo V.D., Gioviale M.C., Bellavia M., Tomasello G., Lo Monte A.I. The mediterranean diet: A history of health. Iran. J. Public Health. 2013;42:449–457.
- 54.Rosato V., Temple N.J., La Vecchia C., Castellan G., Tavani A., Guercio V. Mediterranean diet and cardiovascular disease: A systematic review and meta-analysis of observational studies. Eur. J. Nutr. 2019;58:173–191. doi: 10.1007/s00394-017-1582-0.
- 55.Schwingshackl L., Schwedhelm C., Galbete C., Hoffmann G. Adherence to Mediterranean diet and risk of cancer: An updated systematic review and meta-analysis. Nutrition. 2017;9:63. doi: 10.3390/nu9101063.
- 56.Esposito K., Maiorino M.I., Bellastella G., Chiodini P., Panagiotakos D., Giugliano D. A journey into a Mediterranean diet and type 2 diabetes: A systematic review with meta-analyses. BMJ Open. 2015;5:e008222. doi: 10.1136/bmjopen-2015-008222.
- 57.Petersson S.D., Philippou E. Mediterranean diet, cognitive function, and dementia: A systematic review of the evidence. Adv. Nutr. 2016;7:889–904. doi: 10.3945/an.116.012138.
- 58.Loughrey D.G., Lavecchia S., Brennan S., Lawlor B.A., Kelly M.E. The impact of the Mediterranean diet on the cognitive functioning of healthy older adults: A systematic review and meta-analysis. Adv. Nutr. 2017;8:571–586. doi: 10.3945/an.117.015495.
- 59.Estruch R., Ros E., Salas-Salvado J., Covas M.I., Corella D., Aros F., Gomez-Gracia E., Ruiz-Gutierrez V., Fiol M., Lapetra J., et al. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N. Engl. J. Med. 2018;378:e34. doi: 10.1056/NEJMoa1800389.
- 60.Medina-Remon A., Tresserra-Rimbau A., Pons A., Tur J.A., Martorell M., Ros E., Buil-Cosiales P., Sacanella E., Covas M.I., Corella D., et al. Effects of total dietary polyphenols on plasma nitric oxide and blood pressure in a high cardiovascular risk cohort. The PREDIMED randomized trial. Nutr. Metab. Cardiovasc. Dis. 2015;25:60–67. doi: 10.1016/j.numecd.2014.09.001.
- 61.Sala-Vila A., Romero-Mamani E.S., Gilabert R., Nunez I., de la Torre R., Corella D., Ruiz-Gutierrez V., Lopez-Sabater M.C., Pinto X., Rekondo J., et al. Changes in ultrasound-assessed carotid intima-media thickness and plaque with a Mediterranean diet: A substudy of the PREDIMED trial. Arter. Thromb Vasc. Biol. 2014;34:439–445. doi: 10.1161/ATVBAHA.113.302327.
- 62.Medina-Remon A., Zamora-Ros R., Rotches-Ribalta M., Andres-Lacueva C., Martinez-Gonzalez M.A., Covas M.I., Corella D., Salas-Salvado J., Gomez-Gracia E., Ruiz-Gutierrez V., et al. Total polyphenol excretion and blood pressure in subjects at high cardiovascular risk. Nutr. Metab. Cardiovasc. Dis. 2011;21:323–331. doi: 10.1016/j.numecd.2009.10.019.
- 63.Medina-Remon A., Casas R., Tressserra-Rimbau A., Ros E., Martinez-Gonzalez M.A., Fito M., Corella D., Salas-Salvado J., Lamuela-Raventos R.M., Estruch R. Polyphenol intake from a Mediterranean diet decreases inflammatory biomarkers related to atherosclerosis: A substudy of the PREDIMED trial. Br. J. Clin. Pharmacol. 2017;83:114–128. doi: 10.1111/bcp.12986.
- 64.Sacks F.M., Svetkey L.P., Vollmer W.M., Appel L.J., Bray G.A., Harsha D., Obarzanek E., Conlin P.R., Miller E.R., Simons-Morton D.G., et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N. Engl. J. Med. 2001;344:3–10. doi: 10.1056/NEJM200101043440101.
- 65.Siervo M., Lara J., Chowdhury S., Ashor A., Oggioni C., Mathers J.C. Effects of the Dietary Approach to Stop Hypertension (DASH) diet on cardiovascular risk factors: A systematic review and meta-analysis. Br. J. Nutr. 2015;113:1–15. doi: 10.1017/S0007114514003341.
- 66.Soltani S., Shirani F., Chitsazi M.J., Salehi-Abargouei A. The effect of dietary approaches to stop hypertension (DASH) diet on weight and body composition in adults: A systematic review and meta-analysis of randomized controlled clinical trials. Obes. Rev. 2016;17:442–454. doi: 10.1111/obr.12391.
- 67.Shirani F., Salehi-Abargouei A., Azadbakht L. Effects of Dietary Approaches to Stop Hypertension (DASH) diet on some risk for developing type 2 diabetes: A systematic review and meta-analysis on controlled clinical trials. Nutrients. 2013;29:939–947. doi: 10.1016/j.nut.2012.12.021.
- 68.Chiavaroli L., Viguiliouk E., Nishi S.K., Blanco Mejia S., Rahelic D., Kahleova H., Salas-Salvado J., Kendall C.W., Sievenpiper J.L. DASH dietary pattern and cardiometabolic outcomes: An umbrella review of systematic reviews and meta-analyses. Nutrients. 2019;11:338. doi: 10.3390/nu11020338.
- 69.Salehi-Abargouei A., Maghsoudi Z., Shirani F., Azadbakht L. Effects of Dietary Approaches to Stop Hypertension (DASH)-style diet on fatal or nonfatal cardiovascular diseases--incidence: A systematic review and meta-analysis on observational prospective studies. Nutrients. 2013;29:611–618. doi: 10.1016/j.nut.2012.12.018.
- 70.Asemi Z., Samimi M., Tabassi Z., Esmaillzadeh A. The effect of DASH diet on pregnancy outcomes in gestational diabetes: A randomized controlled clinical trial. Eur. J. Clin. Nutr. 2014;68:490–495. doi: 10.1038/ejcn.2013.296.
- 71.Asghari G., Yuzbashian E., Mirmiran P., Hooshmand F., Najafi R., Azizi F. Dietary Approaches to Stop Hypertension (DASH) dietary pattern is associated with reduced incidence of metabolic syndrome in children and adolescents. J. Pediatr. 2016;174:178–184.e1. doi: 10.1016/j.jpeds.2016.03.077.
- 72.Han S., Middleton P., Shepherd E., Van Ryswyk E., Crowther C.A. Different types of dietary advice for women with gestational diabetes mellitus. Cochrane Database Syst. Rev. 2017;2:Cd009275. doi: 10.1002/14651858.CD009275.pub3.
- 73.van de Rest O., Berendsen A.A., Haveman-Nies A., de Groot L.C. Dietary patterns, cognitive decline, and dementia: A systematic review. Adv. Nutr. 2015;6:154–168. doi: 10.3945/an.114.007617.
- 74.Chen X., Maguire B., Brodaty H., O’Leary F. Dietary patterns and cognitive health in older adults: A systematic review. J. Alzheimers Dis. 2019;67:583–619. doi: 10.3233/JAD-180468.
- 75.Wengreen H., Munger R.G., Cutler A., Quach A., Bowles A., Corcoran C., Tschanz J.T., Norton M.C., Welsh-Bohmer K.A. Prospective study of Dietary Approaches to Stop Hypertension- and Mediterranean-style dietary patterns and age-related cognitive change: The Cache County Study on Memory, Health and Aging. Am. J. Clin. Nutr. 2013;98:1263–1271. doi: 10.3945/ajcn.112.051276.
- 76.Morris M.C., Tangney C.C., Wang Y., Sacks F.M., Barnes L.L., Bennett D.A., Aggarwal N.T. MIND diet slows cognitive decline with aging. Alzheimers Dement. 2015;11:1015–1022. doi: 10.1016/j.jalz.2015.04.011.
- 77.Adamsson V., Reumark A., Fredriksson I.B., Hammarstrom E., Vessby B., Johansson G., Riserus U. Effects of a healthy Nordic diet on cardiovascular risk factors in hypercholesterolaemic subjects: A randomized controlled trial (NORDIET) J. Intern. Med. 2011;269:150–159. doi: 10.1111/j.1365-2796.2010.02290.x.
- 78.Jensen J.D., Poulsen S.K. The new Nordic diet–consumer expenditures and economic incentives estimated from a controlled intervention. BMC Public Health. 2013;13:1114. doi: 10.1186/1471-2458-13-1114.
- 79.Mithril C., Dragsted L.O., Meyer C., Blauert E., Holt M.K., Astrup A. Guidelines for the New Nordic Diet. Public Health Nutr. 2012;15:1941–1947. doi: 10.1017/S136898001100351X.
- 80.Nordic Nutrition Recommendations 2012: Integral Nutrition and Physical Activity. Nordic Council of Ministers; Copenhagen, Denmark: 2014.
- 81.Uusitupa M., Hermansen K., Savolainen M.J., Schwab U., Kolehmainen M., Brader L., Mortensen L.S., Cloetens L., Johansson-Persson A., Onning G., et al. Effects of an isocaloric healthy Nordic diet on insulin sensitivity, lipid profile and inflammation markers in metabolic syndrome—A randomized study (SYSDIET) J. Intern. Med. 2013;274:52–66. doi: 10.1111/joim.12044.
- 82.Brader L., Uusitupa M., Dragsted L.O., Hermansen K. Effects of an isocaloric healthy Nordic diet on ambulatory blood pressure in metabolic syndrome: A randomized SYSDIET sub-study. Eur. J. Clin. Nutr. 2014;68:57–63. doi: 10.1038/ejcn.2013.192.
- 83.Poulsen S.K., Due A., Jordy A.B., Kiens B., Stark K.D., Stender S., Holst C., Astrup A., Larsen T.M. Health effect of the New Nordic Diet in adults with increased waist circumference: A 6-mo randomized controlled trial. Am. J. Clin. Nutr. 2014;99:35–45. doi: 10.3945/ajcn.113.069393.
- 84.Marklund M., Magnusdottir O.K., Rosqvist F., Cloetens L., Landberg R., Kolehmainen M., Brader L., Hermansen K., Poutanen K.S., Herzig K.H., et al. A dietary biomarker approach captures compliance and cardiometabolic effects of a healthy Nordic diet in individuals with metabolic syndrome. J. Nutr. 2014;144:1642–1649. doi: 10.3945/jn.114.193771.
- 85.Sorensen L.B., Damsgaard C.T., Dalskov S.M., Petersen R.A., Egelund N., Dyssegaard C.B., Stark K.D., Andersen R., Tetens I., Astrup A., et al. Diet-induced changes in iron and n-3 fatty acid status and associations with cognitive performance in 8–11-year-old Danish children: Secondary analyses of the Optimal Well-Being, Development and Health for Danish Children through a Healthy New Nordic Diet School Meal Study. Br. J. Nutr. 2015;114:1623–1637. doi: 10.1017/S0007114515003323.
- 86.Akesson A., Andersen L.F., Kristjansdottir A.G., Roos E., Trolle E., Voutilainen E., Wirfalt E. Health effects associated with foods characteristic of the Nordic diet: A systematic literature review. Food Nutr. Res. 2013;57 doi: 10.3402/fnr.v57i0.22790.
- 87.Kim S., Kim M., Lee M., Park Y., Lee H., Kang S., Lee H., Lee K., Yang H., Kim M., et al. Korean diet: Characteristics and historical background. J. Ethn. Foods. 2016;3:26–31. doi: 10.1016/j.jef.2016.03.002.
- 88.Lee K.W., Cho M.S. The traditional Korean dietary pattern is associated with decreased risk of metabolic syndrome: Findings from the Korean National Health and Nutrition Examination Survey, 1998–2009. J. Med. Food. 2014;17:43–56. doi: 10.1089/jmf.2013.3049.
- 89.Jung S.J., Park S.H., Choi E.K., Cha Y.S., Cho B.H., Kim Y.G., Kim M.G., Song W.O., Park T.S., Ko J.K., et al. Beneficial effects of Korean traditional diets in hypertensive and type 2 diabetic patients. J. Med. Food. 2014;17:161–171. doi: 10.1089/jmf.2013.3042.
- 90.Ma G. Food, eating behavior and culture in Chinese society. J. Ethn. Foods. 2015;2:195–199. doi: 10.1016/j.jef.2015.11.004.
- 91.Introducing the Updated Asian Diet Pyramid. [(accessed on 29 May 2019)]; Available online: https://oldwayspt.org/blog/introducing-updated-asian-diet-pyramid.
- 92.Wan Y., Wang F., Yuan J., Li J., Jiang D., Zhang J., Huang T., Zheng J., Mann J., Li D. Effects of macronutrient distribution on weight and related cardiometabolic profile in healthy non-obese Chinese: A 6-month, randomized controlled-feeding trial. EBioMedicine. 2017;22:200–207. doi: 10.1016/j.ebiom.2017.06.017.
- 93.Leonetti F., Liguori A., Petti F., Rughini S., Silli L., Liguori S., Bangrazi S. Effects of basic traditional Chinese diet on body mass index, lean body mass, and eating and hunger behaviours in overweight or obese individuals. J. Tradit Chin. Med. 2016;36:456–463. doi: 10.1016/S0254-6272(16)30062-0.
- 94.Liguori A., Petti F., Rughini S., Silli L., Asprino R., Maglio C., Leonetti F. Effect of a basic Chinese traditional diet in overweight patients. J. Tradit Chin. Med. 2013;33:322–324. doi: 10.1016/S0254-6272(13)60173-9.
- 95.Gabriel A.S., Ninomiya K., Uneyama H. The role of the Japanese traditional diet in healthy and sustainable dietary patterns around the world. Nutrients. 2018;10:173. doi: 10.3390/nu10020173.
- 96.Niu K., Momma H., Kobayashi Y., Guan L., Chujo M., Otomo A., Ouchi E., Nagatomi R. The traditional Japanese dietary pattern and longitudinal changes in cardiovascular disease risk factors in apparently healthy Japanese adults. Eur. J. Nutr. 2016;55:267–279. doi: 10.1007/s00394-015-0844-y.
- 97.Htun N.C., Suga H., Imai S., Shimizu W., Takimoto H. Food intake patterns and cardiovascular risk factors in Japanese adults: Analyses from the 2012 National Health and nutrition survey, Japan. Nutr. J. 2017;16:61. doi: 10.1186/s12937-017-0284-z.
- 98.Turconi G.T., Nucera A., Roggi C., Cena H. Food consumption and diet cost: A northern Italian survey. Italy J. Food Sci. 2010;22:352–358.
- 99.Bourassa M.W., Osendarp S.J.M., Adu-Afarwuah S., Ahmed S., Ajello C., Bergeron G., Black R., Christian P., Cousens S., de Pee S., et al. Review of the evidence regarding the use of antenatal multiple micronutrient supplementation in low- and middle-income countries. Ann. N. Y. Acad. Sci. 2019;1444:6–21. doi: 10.1111/nyas.14121.
- 100.Swed-Tobia R., Haj A., Militianu D., Eshach O., Ravid S., Weiss R., Aviel Y.B. Highly selective eating in autism spectrum disorder leading to scurvy: A series of three patients. Pediatr. Neurol. 2019;94:61–63. doi: 10.1016/j.pediatrneurol.2018.12.011.
- 101.National Institute of Health Calcium Fact Sheet for Health Professionals. [(accessed on 12 February 2019)]; Available online: https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional/
- 102.African Heritage Diet. [(accessed on 6 January 2020)]; Available online: https://oldwayspt.org/traditional-diets/african-heritage-diet.
Original written by from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7071223/
